在学习A-level化学时,要学会总结题目中涉及到的化学反应类型,一点一滴地积累,从而做到积少成多。下面就是在A-level有机化学中涉及到的化学反应。
1. Alkane à chloroalkane
reagents: Cl2
conditions: UV light
mechanism: free radical substitution
equation: RH + Cl2 à RCl + HCl
2. Alkene à polyalkene
Conditions: low T, high p.
Equation:
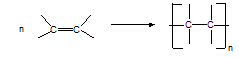
Type of reaction: addition polymerisation (free radical)
3. Alkene à bromoalkane
Reagent: HX(g)
Conditions: room T
Equation:
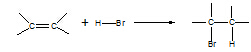
Type of reaction: electrophilic addition
4. Alkene à dibromoalkane
Reagent: Br2 in water or in an organic solvent
Conditions: room T
Equation:
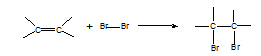
Mechanism: electrophilic addition
5. Alkene à alkylhydrogensulphate
Reagent: concentrated sulphuric acid
Conditions: cold
Equation:
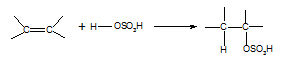
Mechanism: electrophilic addition
6. Alkylhydrogensulphate à alcohol
Reagent: water
Conditions: warm
Equation:
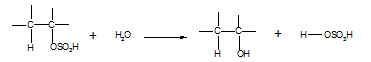
Type of reaction: hydrolysis
7. Alkene à alcohol
Reagent: steam
Conditions: 300 oC, 60 atm, H3PO4 catalyst
Equation:
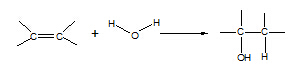
Type of reaction: hydration
8. Haloalkane à alcohol
Reagent: NaOH(aq) or KOH(aq)
Conditions: warm under reflux
Equation: R-X + OH- à R-OH + X-
Type of reaction: nucleophilic substitution
9. Haloalkane à nitrile
Reagent: KCN in aqueous ethanol
Conditions: boil under reflux
Equation: R-X + CN- à R-CN + X-
Type of reaction: nucleophilic substitution
10. Haloalkane à Amine
Reagent: ammonia in ethanol in a sealed tube
Conditions: heat
Equation: R-X + 2NH3 à R-NH2 + NH4X
Type of reaction: nucleophilic substitution
11. Haloalkane à alkene
Reagent: KOH in ethanol
Conditions: heat
Equation:
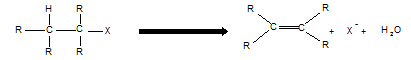
Type of reaction: elimination
12. Primary alcohol à aldehyde
Reagent: potassium dichromate and dilute sulphuric acid
Conditions: warm, distillation
Equation: RCH2OH + [O] à RCHO + H2O
Type of reaction: mild oxidation
13. Secondary alcohol à ketone
Reagent: potassium dichromate and dilute sulphuric acid
Conditions: heat, distillation
Equation: R1CH(OH)R2 + [O] à R1COR2 + H2O
Type of reaction: oxidation
14. aldehyde à carboxylic acid
Reagent: potassium dichromate and dilute sulphuric acid
Conditions: heat, reflux
Equation: R-CHO + [O] à R-COOH
Type of reaction: oxidation
15. Alcohols à alkenes
Reagent: concentrated sulphuric acid
Conditions: heat
Equation:
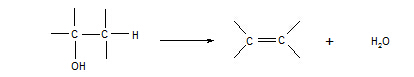
Type of reaction: elimination
16. glucose à ethanol
reagent: yeast
conditions: 35 – 55 oC, no air
equation: C6H12O6 à 2C2H5OH + 2CO2
type of reaction: fermentation




